
When a patient presents with symptoms of premature rupture of membranes (PROM), she is counting on you for accurate and reliable results. The right decision for her and her baby starts with AmniSure. See why our clinicians include AmniSure as part of their clinical assessment of PROM – see why they love AmniSure.
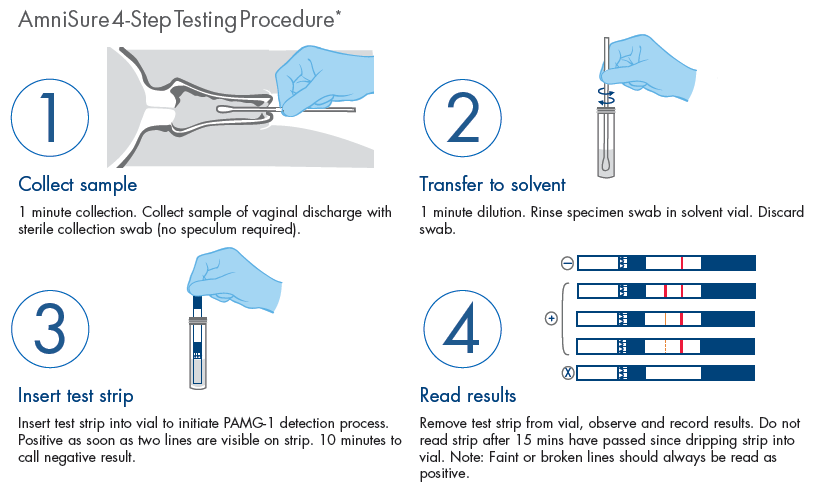 AmniSure is a rapid, non-instrumented, qualitative immunoassay to aid in the detection of ROM (rupture of [fetal] membranes). AmniSure is 98.9% sensitive and 98.1% specific when used as part of the overall clinical assessment.
AmniSure is a rapid, non-instrumented, qualitative immunoassay to aid in the detection of ROM (rupture of [fetal] membranes). AmniSure is 98.9% sensitive and 98.1% specific when used as part of the overall clinical assessment.
Accurate Insights
Think about the number of unnecessary treatments you may be able to avoid with the most accurate ROM Test. AmniSure delivers 98% specificity to aid in the detection of ROM.
| AmniSure ROM Test (1) | ROM Plus Test (2) | Actim PROM (3) |
|
|---|---|---|---|
| Amniotic fluid biomarker(s) detected | PAMG-1 | IGFBP-1 (PP12) & AFP | IGFBP-1 |
| Overall specificity | 100% without speculum | 75% without speculum | 91.0% without speculum 86.4% with speculum |
| Overall sensitivity | 98.9% without speculum | 99.1% without speculum | 90.1% without speculum 95.5% with speculum |
Reliable in blood
With up to 30% of suspected ROM patients presenting with vaginal bleeding (4), it is critical to have a ROM test that is reliable, even in the presence of blood. In a published study, AmniSure was shown to be more accurate and readable in the presence of vaginal bleeding (4).
| Test | Sensivity | Specificity | Overall accuracy | PPV | NPV | Tests non-evaluable |
|---|---|---|---|---|---|---|
| AmniSure | 97.8% | 91.5% | 95.3% | 94.6% | 96.4% | 3 (1.9%) |
| Actim PROM | 91.0% | 75.0% | 84.3% | 83.5% | 85.7% | 17 (11.3%) |
 No age limit. No speculum required.
No age limit. No speculum required.
Improve your OB clinical assessment of ROM. AmniSure does not have a gestational age limit and speculum is not required when collecting a sample for AmniSure.
Evidence-based practice
AmniSure is cited in over 20 peer-reviewed studies and is used in birthing hospitals in almost 80 countries worldwide when diagnosing ROM.
 The only test that stacks up to the gold standard
The only test that stacks up to the gold standard
AmniSure is the only ROM diagnostic test that was compared to the gold standard, indigo carmine amnio-dye infusion. The published study shows AmniSure has a ~99% correlation with the indigo carmine test (5).
